by Robert W. Chandler, M.D., M.B.A. - Team 5
At the launch of widespread mass
inoculation of the public with Pfizer’s mRNA vaccine, BNT162b2, media,
physicians’ spokespeople, and government officials communicated widely
that the injected drug would be retained at the injection site muscle
tissue and in local lymph nodes. The components were supposed to be
metabolized in a day or so, leaving only induced SARS CoV-2 Spike
antigen to evoke a therapeutic immune response. A short pulse of drug
effect would be followed, they claimed, by limited production of Spike
antigen.
[
https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M2_26_pharmkin-tabulated-summary.pdf] and the heavily redacted report “
R&D
STUDY REPORT No. R-20-0072 – EXPRESSION OF LUCIFERASE-ENCODING MODERNA
AFTER I.M. APPLICATION OF GMPREADY ACUITAS LIPID NANOPARTICLE
FORMULATION “[
https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_R-20-0072.pdf] — all examine tissue distribution of Pfizer’s mRNA vaccine BNT162b2. These documents will be addressed in this report.
Pfizer Study 185350,” Final
Report: A Tissue Distribution Study of a [3H]-Labelled Lipid
Nanoparticle-mRNA Formulation Containing ALC-0315 and ALC-0159 Following
Intramuscular Administration in Wistar Han Rat”,
is one of 21 preclinical Prizer studies involving mice, rats and rhesus
macaque non-human primates. Study No. 185350 (Sponsor Reference
ALC-NC-0552) was summarized in Pfizer’s “2.4 Nonclinical Overview” and
was separately published as a Final Report dated September 24, 2020.
Contained in that document is the following identification of the source:
Test Facility Study No. 185350 REDACTED
SPONSOR: Acuitas,
6190 Agronomy Road,
Ste. 402,
Vancouver, V6T 1Z3 Canada
Sponsor Reference No. ALC-NC-0552
This study was made up of 42 male
and 21 female Wistar Han rats. These rats were injected with 50 or 100
micrograms of BNT162b2 mRNA/LNP (lipid nanoparticle) product labelled
with a radioactive tracer material, 3H.
Then the rats were sacrificed at intervals of 0.25 hours (15 minutes); 1
hour; 2 hours; 4 hours; 8 hours; and then at 1 and 2 days.
The results of 21 male and 21 female sacrificed rats are presented.
The 100-microgram dose was associated
with loss of weight and apparent toxicity in two animals.
Unfortunately, the full results of the 100-microgram dose were not
presented at all. [https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_185350.pdf, p. 11.]
This is very important. The 100 microgram dose was considered too
toxic to continue to use in the experiment, so the dosage was cut in
half. 100 micrograms is the amount in the Moderna injections.
The 50 microgram dose was not safe. One female rat in the 50-microgram dose exhibited piloerection and hunched posture. [https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_185350.pdf, p.19.]
The injection did not stay at the injection site, as we were promised
it would. Rather, following injection, the drug was persistent at the
injection site, with a third of the dose remaining in muscle tissue for
two days in males, and a sixth of the dose remained in females for the
same duration.
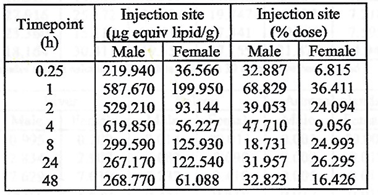
But it did not all stay in the deltoid muscle. From the injection site
in the deltoid muscle, mRNA/ Lipid Nanoparticles appeared in blood and
plasma fifteen minutes after injection and persisted for the entire
duration of the two-day study.
On page 20 of “Final Report: A Tissue Distribution Study of a
[3H]-Labelled Lipid Nanoparticle-mRNA Formulation Containing ALC-0315
and ALC-0159 Following Intramuscular Administration in Wistar Han Rat,”
the authors note that widespread distribution to “most tissues” occurs
by the time of first analysis at 15 minutes after injection.
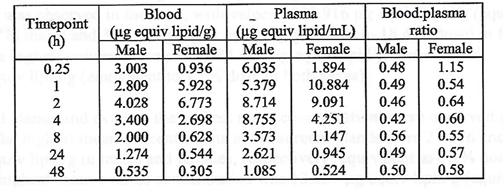
There was greater accumulation in blood when compared to plasma, and
males generally had higher concentrations than females with lower blood
to plasma ratios. No explanation for these differences was offered.
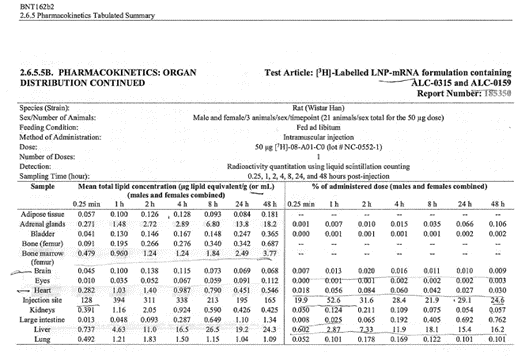
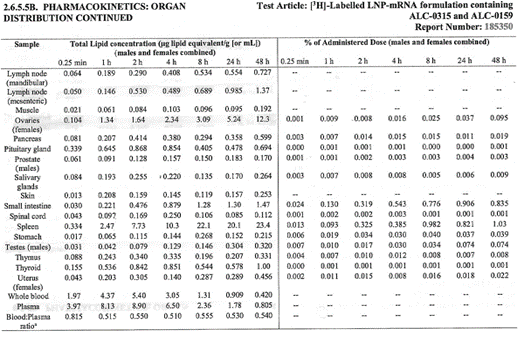
The major tissues that contained the drug concentration, aside from
muscle at the injection site, were identified as being the liver,
spleen, adrenal glands, and ovaries. The drug persisted in tissues
throughout the duration of the study. The meaning and potential
implications of the persistence in tissues was not addressed. [https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_185350.pdf, p. 21.]
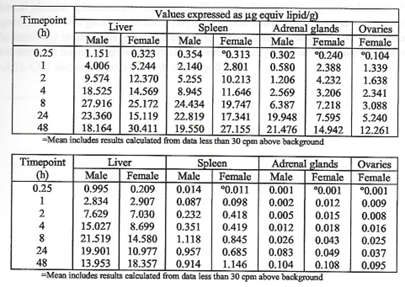 Top: highest mean concentrations. Bottom: equivalent % dose.
Top: highest mean concentrations. Bottom: equivalent % dose.
The next two tables present the overall tissue distribution data from
this study. It is reasonable to conclude, thus, that BNT162b2 is
distributed throughout the body and persists for at least two days, the
duration of the study. [
https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_185350.pdf,
pp. 7-8.] Tissue specimens were harvested but, unfortunately, no
microscopic analysis of these specimens is presented at all, so
potential damage to various organs was not evaluated.
A separate pharmacokinetic study, “PF-07302048,” looked at the
persistence of the LNP (lipid nanoparticle) transport vessel with a test
mRNA inside consisting of LNP coating wrapped around Luciferase mRNA,
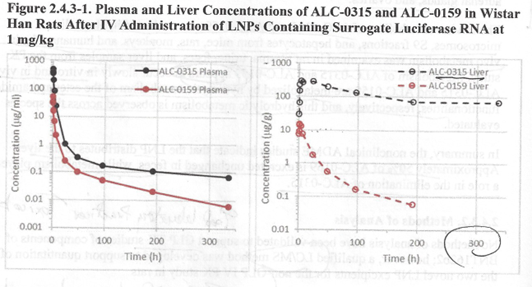
Figure 2.4.3-1 below. [“R&D STUDY REPORT No. R-20-0072 –
EXPRESSION OF LUCIFERASE-ENCODING MODRNA AFTER I.M. APPLICATION OF
GMPREADY ACUITAS LIPID NANOPARTICLE FORMULATION”, https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_R-20-0072.pdf.]
The object of this study was to follow the LNP vessel in plasma and
liver, and then measure transcription of mRNA inside target organs to
validate the delivery model using the bioluminescent properties of
Luciferase to identify transcription of the mRNA in target tissues. [
https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M4_4223_R-20-0072.pdf
From this study, we learn that the two measured components of the lipid
nanoparticle coating, ALC-0315 [(4-hydroxybutyl) azanediyl]di(hexane-6,
1-diyl) bis (2-hexyldecanooate)] and ALC-0159 (2-[2-(polyethylene
glycol)-2000]-N, N-ditetradecylacetamide) are detectable in plasma after
300 hours – that is to say, 12.5 days – which fact raises the issue of
how long the contents of the LNP vessel with the mRNA inside persists,
and what the implications are of prolonged occupation of host cells by
this material. In this study, the BNT162b2 was injected intravenously,
accelerating the dissemination of drug. [
2.4 NONCLINICAL OVERVIEW,
https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M2_24_nonclinical-overview.pdf, p.16.]
Figure 1: From R&D STUDY REPORT No. R-20-0072 – EXPRESSION OF
LUCIFERASE-ENCODING MODRNA AFTER I.M. APPLICATION OF GMPREADY ACUITAS
LIPID NANOPARTICLE FORMULATION.
T
his study of the biodistribution of the LNP coating containing
Luciferase mRNA found that not only was the mRNA transcribed, but the
LNP “vessel” components ALC-0315 and ALC-0159 were retained in the liver
and in the plasma for at least 12.5 days. The fate of the Luciferase
mRNA was not discussed.With respect to degradation of the mRNA component, we learn from “2.4 Nonclinical Overview”
that Pfizer/Acuitas did not study at all the degradation of the
synthetic mRNA in BNT162b2. Similarly, there was no analysis by Pfizer
of protein products from BNT162b2 provided. [https://www.phmpt.org/wp-content/uploads/2022/03/125742_S1_M2_24_nonclinical-overview.pdf, p.20.]

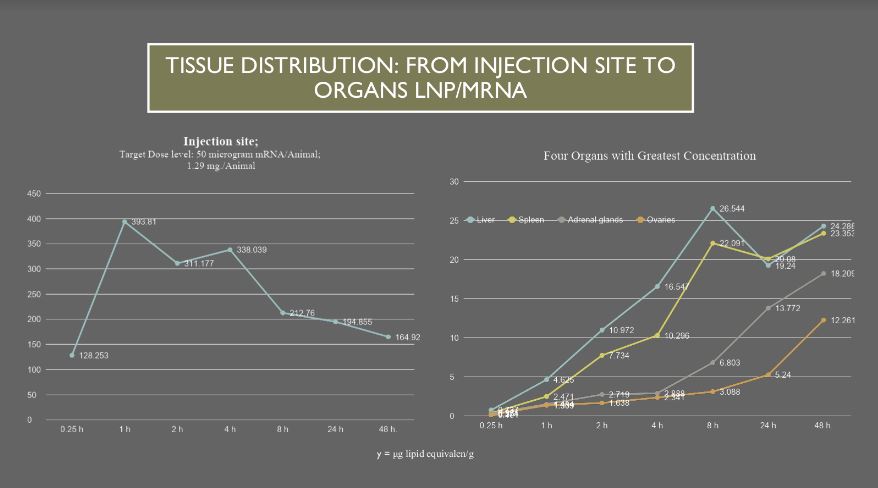
Liver, Spleen, Adrenal glands and Ovaries take up increasing
amounts of drug compared with other organs as drug is transported from
the injection site by blood and plasma. These data were generated during
the 48 hours after injection and these four organs were still
accumulating the transferred drug as the experiment ended. Looking at
the tissue concentration shown on the y axis, there is 165 microgram
lipid equivalent/g drug remaining at the injection site compared with 24
microgram lipid equivalent/g in the liver. This indicates that there
may be continued transport from the injection site by blood and plasma
well beyond 48 hours when the last animals were sacrificed.
The remaining plots show patterns of drug distribution by blood and
plasma and accumulation in organs. Page 1 lists the table of values
following injection of radioactive labeling of the LNP component of
BNT162b2. Pages 2-16 are plots of drug in Adrenal glands, Bone marrow,
Brain, Heart, Kidneys, Liver, Lung, Lymph nodes mandibular and
mesenteric, Ovaries, Spleen, Thyroid, Blood and Plasma, Injection site,
mandibular lymph nodes vs. mesenteric, and organs with greatest
concentrations of the drug.
BNT162b2 has lipid nanoparticles that may have adverse effects on
organs and tissues from the lipid component as well as from the mRNA and
Spike proteins produced by the mRNA.
Graphical representation of the Pfizer data set allows the reader
to get a better understanding of how BNT162b2 flows from the injection
site to body organs.
For example, this chart is a plot of tissue concentration first
rising steeply at the injection site, left chart, then peaks and
declines as the organ systems accumulate concentrations of drug more
slowly early then rise over time, right chart.
The final chart on page 19 contrasts ovaries vs testes with
approximately 38 times more drug concentration of drug in ovaries. More
work is needed to see if there is a connection between menstrual cycle
changes as have been reported by Lee, et. al. and elsewhere.
Similarly, although the data is incomplete
with known outcome in only 27 of 270 pregnancy cases as reported in
Pfizer document 5.3.6, the first three months of Adverse Events
reporting following widespread release after an Emergency Use
Authorization was given by the FDA. What data does exist is
disconcerting.
Several serious questions are raised by these results:
- How long does the BNT162b2 mRNA persist in human tissues? Where does
it go in the host cell? How long does it persist inside the cell? What
proteins does it produce, and for how long?
- Is there any possibility that the BNT162b2 mRNA can be transcribed
into DNA, then incorporate into the host genome? If this happens what
are the implications?
- What are the toxicities from the lipid nanoparticle coating?
- Was Pfizer obligated to answer these questions prior to human testing?
- Doesn’t proper informed consent require answers to these questions?
Fortunately, answers to these important questions are beginning to appear:
1a. Duration of mRNA in tissues:
In a July 19, 2022, article, the essayist Joomi reviews the topic of
how long BNT162 b2 containing mRNA stabilized by a synthetic nucleotide
1N-methyl pseudouridine persists in human tissues. [https://joomi.substack.com/p/were-still-being-misled-about-how?r=chkp3&s=r&utm_campaign=post&utm_medium=web]
A January 2022 human lymph node biopsy study from Stanford University
found that the mRNA from both Pfizer and Moderna persists for at least
two months, which was the duration of the study. [https://www.cell.com/action/showPdf?pii=S0092-8674%2822%2900076-9]
1b. Proteins produced from BNT162b2 mRNA:
Spike protein is produced after the mRNA is transcribed, and has been
found in vivo for at least four months after inoculation. [https://joomi.substack.com/p/were-still-being-misled-about-how?r=chkp3&s=r&utm_campaign=post&utm_medium=web]
Proteins transcribed from the mRNA have not been completely
characterized yet. SARS-CoV-2-like Spike protein has been identified as
long as four months after inoculation with LNP/mRNA in human exosomes.
Toxicity of Spike protein has been described and is reviewed in the
essay “We’re still being misled about how long the mRNA vaccines last in
the body.” [https://joomi.substack.com/p/were-still-being-misled-about-how?r=chkp3&s=r&utm_campaign=post&utm_medium=web]
2. What is the fate of BNT162b2 mRNA?
We were informed that “RNA is required for protein synthesis, does
not integrate into the genome, is transiently expressed, and is
metabolized and is eliminated by the body’s natural mechanisms and,
therefore is considered safe.” [Alberer, M. et al. Safety and
immunogenicity of a mRNA rabies vaccine in healthy adults: an
open-label, non-randomized, prospective, first-in-human phase 1 clinical
trial. Lancet 90, 1511-1520 (2017).] [Sahin, U. e al.
Personalized RNA mutanome vaccines mobilize poly-specific therapeutic
immunity against cancer. Nature 547, 222-226 (2017).]
However, Alden, et. al., reporting in Current Issues in Molecular Biology 2022, 44, 1115-1126, found BNT162b2 mRNA is reverse transcribed into host DNA beginning six hours after contact with BNT162b2:
“In the BNT162b2 toxicity report, no genotoxicity nor
carcinogenicity studies have been provided. Our study shows that
BNT162b2 can be reverse transcribed to DNA in liver cell line Huh7, and
this may give rise to the concern if BNT162b2-derived DNA may be
integrated into the host genome and affect the integrity of genomic DNA,
which may potentially mediate genotoxic side effects. At this stage, we
do not know if DNA reverse transcribed from BNT162b2 is integrated into
the cell genome. Further studies are needed to demonstrate the effect
of BNT162b2 on genomic integrity, including whole genome sequencing of
cells exposed to BNT162b2, as well as tissues from human subjects who
received BNT162b2 vaccination.” [
https://www.mdpi.com/1467-3045/44/3/73/htmThis study did not identify DNA transcribed from BNT162b2 mRNA in the host genome following transcription.
However, Zhang et. al., working at Massachusetts Institute of
Technology, demonstrated fragments of SARS-CoV-2 mRNA integrated in host
DNA in “Reverse-transcribed SARS-CoV-2 RNA can integrate into the
genome of cultured human cells and can be expressed in patient-derived
tissues,” published in 2021 in PNAS, vol. 118, no. 21:
“We show here that SARS-CoV-2 RNA can be
reverse-transcribed and integrated into the genome of the infected cell
and be expressed as chimeric transcripts fusing viral with cellular
sequences. Importantly, such chimeric transcripts are detected in
patient-derived tissues.” [https://www.pnas.org/doi/10.1073/pnas.2105968118]
So, scientists are getting close to knowing whether BNT162b2, with
its synthetic mRNA, is translated into host DNA and is now a permanent
part of human genetic material. If so, the next step is to determine
what the implications are.
3. What are the toxicities from the lipid nanoparticle coating?
More research is required to understand the implications of LNP
concentration in various organ tissues. It is thought that the PEG
component (the polyethylene glycol that coats the LNP) is responsible
for anaphylaxis, an often rapid-onset major physiologic event that
requires emergency treatment.
4. Was Pfizer obligated to answer these questions prior to human testing?
5. Doesn’t proper informed consent require answers to these questions?
The answers to questions 4 and 5 are “yes,” and the reasons should be
obvious now. Basic information about functioning of this mRNA product,
BNT162b2, was not known at the time of mass inoculation; and, therefore,
a proper risk, benefits and complications discussion was compromised by
lack of information. Informed consent is not possible in such a
situation.
In conclusion, many negatively consequential shortcuts were made in the development of BNT162b2.
Many omissions in basic research evaluation of BNT162b2 were kept
hidden, and there was outright misinformation regarding some of the work
that was done.
Assumptions rather than actual research to determine where BNT162b2
goes, what it does, and how long it lasts were made that proved to be
false and constitute intentional mis/dis/mal information. We were told
that the prodrug, BNT162b2, consisting of a lipid nanoparticle coating
of synthetic messenger ribonucleic acid (modRNA), would be deposited in
muscle tissue at the injection site and would be migrate to local
lymphatics prior to rapid degradation producing Spike antigens for a
limited period of time that would produce a desired immune response.
However, Pfizer in its very early Phase 1 trial with mice, rats, and
rhesus non-human primates learned that the LNP/mRNA is rapidly
disseminated throughout the body and remained in tissues for as long as
it was studied, 48 hours for BNT162b2 and 12.5 days for the
LNP/Luciferase mRNA test product.
No effort was expended to determine what proteins are produced by the
modRNA, what their physiological actions are and how long they are
produced as well as what toxicities and adverse events might be
anticipated with widespread usage of the LNP/mRNA prodrug.
FOIA requests for internal documents from federal health care
agencies, independent review board members, approximately 140 clinical
investigators and Pfizer personnel should be made.
Billions of doses were administered to billions of p
eople. The scale of this potentially massive medical misstep is large.
Ten months to develop novel gene therapy for a novel virus is well
short of the five to 10 years usually required to develop, test and
refine such a product. After billions of doses have been given to
children and adults around the world, possibly altering the course of
human evolution, the public is now seeing the unfortunate consequences
of cutting corners.
This Report was written exclusively for
DailyClout by the Members of the War Room / DailyClout Pfizer Documents
Research Volunteers.
It should not be copied or republished without permission from DailyClout or a full credit and link to DailyClout.io