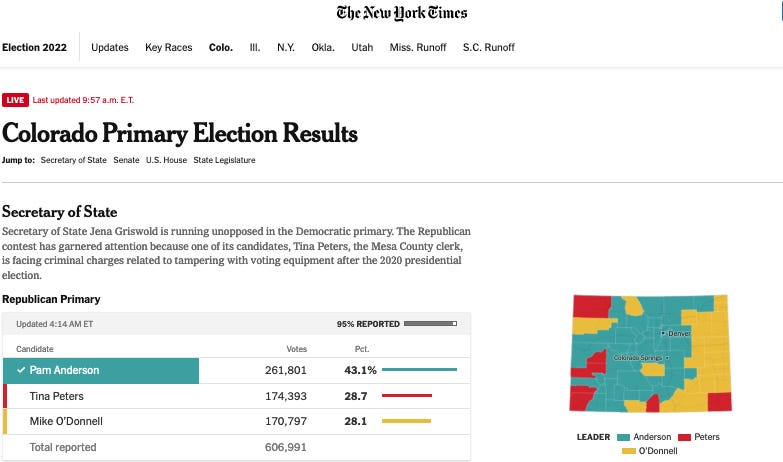
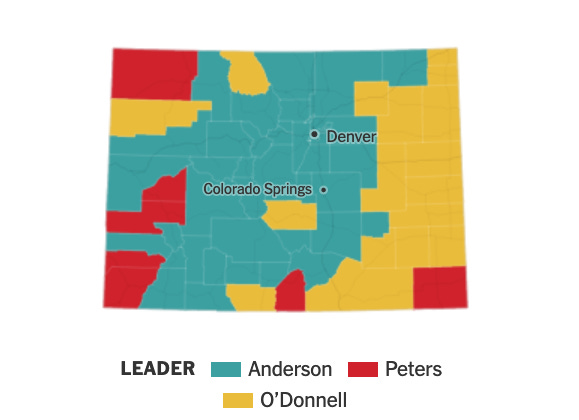
Article by Jerome R. Corsi, Ph.D. republished from
ClimateDepot.com).
This article may contain statements that reflect the opinion of the author
(
Natural News)
Thomas Allmendinger, an independent Swiss physicist, has conducted a
series of experiments published in peer-reviewed scientific journals
that call into question the physical principles of the greenhouse gas
theory. Thomas Allmendinger, an independent scholar educated at the
Swiss Federal Institute of Technology in Zurich, has dared to challenge the conventional politically correct climate
dictum that CO2 is a greenhouse gas with unique global warming
properties. In a series of articles published in scientific journals, Allmendinger has argued that his experimental research with the thermal
absorption of infrared radiation (IR) has proved that “atmospheric trace
gases such as carbon dioxide do not have any influence on the climate.”
The theory of greenhouse gases traces back to an 1827 publication by French physicist Jean Baptiste Joseph Fourier entitled “Mémoire sur les temperature du globe Terrestre ed de espaces planétaires,” [in English: “Memoir on the temperatures of the terrestrial globe and planetary spaces.”]
Like many metaphors in science, the greenhouse image is deceptive in
that it suggests certain gases like water vapor and CO2 act as a shield
preventing infrared radiation (IR) emitted by the sun and absorbed by
Earth from escaping at night into outer space.
A source as supposedly authoritative as NASA still elaborates on the greenhouse gas effect as follows:
“A real greenhouse is made of glass, which lets visible sunlight through
from the outside. This light gets absorbed by all the materials
inside, and the warmed surfaces radiate infrared light, sometimes called
“heat rays,” back. But the glass, although transparent to visible
light, acts as a partial shield to the infrared light. So, some of this
infrared radiation, or heat, gets trapped inside. The result is that
everything inside the greenhouse, including the air, becomes warmers.”
This NASA description is reminiscent of a gardening website that
explains the complicated physics in these simplistic terms: “While the
sunlight gets in, the heat cannot get out,” a description that
attributes the greenhouse effect to heat convection. But rather than
blocking IR from escaping the glazed undercoating of greenhouse gas,
atmospheric greenhouse gases “shield” against the escape of IR to outer
space by absorbing the escaping IR energy. Conventional IR physics
going back some 200 years relies predominantly on spectrographic
analysis, from which is derived the presumption that only dipole gases like water vapor or CO2 can absorb infrared radiation. Most of the atmosphere is composed of oxygen (O2) and nitrogen (N2), neither of which are dipole gases.
In 2016, Allmendinger published a paper entitled “The thermal
behavior of gases under the influence of infrared radiation” in the
International Journal of Physical Sciences.
Here Allmendinger addressed the conventional wisdom that “any
IR-activity of molecules or atoms requires a shift of the electric
dipole moment, so that two-atomic homo-nuclear molecules (like O2 or N2)
are always IR-active.” Allmendinger insisted this proposition “must be
regarded as a theorem and not as a principal natural law” because
“numerous examples of nonpolar substances are known where an interaction
with electromagnetic radiation occurs, e.g., at halogens where even
colored and thus visible light is absorbed.” What Allmendinger found
surprising was physical scientists had relied almost entirely on
spectrographic analysis to measure the molecular absorption of IR energy
by gases. He stressed that “apparently no thermal measurements have
been made of gases in the presence of IR-radiation, particularly of
sunlight,” even though the primary climate concern with greenhouse gases
involves the thermal absorption of IR energy.
In 2017, Allmendinger published an article entitled “The Refutation of the Climate Greenhouse Theory: A Hopeful Alternative” in Environmental Pollution and Climate Change.
Allmendinger argued for the need to measure thermal absorption to
determine if the spectrographic analysis had failed to detect molecular
activity that absorbed IR energy in non-dipole atmospheric gasses. In
highly technical language, he explained:
“As we know today, photometric absorption is accompanied
by the (quantized) excitation of electrons being followed by a light
emission, due to the back-jumping of the excited electrons into the
ground state. This electronic jumping maybe–but needs not be–associated
with vibrations or rotations of the nuclei in the molecule. In solid
bodies, and to a certain extent also in fluid media, these vibrations or
rotations are not independent but coupled. However, in gases they are
widely independent since the molecules or atoms are moving around
obeying statistical laws, whereby their mean kinetic translational
energy is proportional to their absolute temperature.”
He continued:
“Nevertheless, in the case of an electronic excitation a part of the
vibration or rotation energy may be converted into kinetic energy, and
thus in sensible heat, but the fractional amount of this concerted
energy is not a priori theoretically derivable but must be determined
experimentally. Inversely, part of the kinetic heat energy may be
converted into molecular or atomic vibration energy.”
Allmendinger concluded:
“Thus, in gases two kinds of energy are involved:
“internal” energy being related to intramolecular motions, and
“external” energy being related to intermolecular motions. The first
kind is subject of the quantum mechanics, while the second kind is
subject of the kinetic gas theory. As a consequence, photometric or
spectroscopic measurements cannot deliver quantitative information about
the warming-up of gases due to thermal or other infrared radiation,
while such measurements never have been made so far.”
Allmendinger constructed an experimental apparatus that enabled him
to measure the IR thermal absorption (rather than the spectrographic
light wave absorption) of atmospheric gases, including CO2, O2, N2, and
argon (Ar).
In a 2018 article entitled “The Real Cause of Global Warming and Its Consequences on Climate,” published in the SciFed Journal of Global Warming, Allmendinger
summed up his experimental findings. Allmendinger’s thermal
measurements concluded that “any gas absorbs IR—even noble gases do so
[like Ar]— being warmed up to a limiting temperature which is achieved
when the absorption power is equal to the emission power of the warmed
gas.” He continued: “It could be theoretically demonstrated that the
emission power of a gas is related to the frequency of their particles
(atoms or molecules) and thus to their size.”
Allmendinger’s experimental tests found no significant differences
between the IR absorption capabilities of CO2, O2, N2, or Ar when
thermal absorption was measured instead of spectrographic wave
absorption. “As a consequence, a ‘greenhouse effect’ does not really
exist, at least not related to trace gases such as carbon dioxide.”
The global warming orthodox scientific community has rejected Allmendinger’s work as utter nonsense, arguing that he “is currently not affiliated with any reputable research institute or university.” Yet, Thomas Kuhn, in his highly influential 1962 book The Structure of Scientific Revolutions, reminded
us that scientific paradigm shifts involve revolutions, in which new,
competing theories appear first as “heresies.” Challenges to scientific
orthodoxy must fight their way to acceptance against a legion of
established opponents who have invested careers basing their global
warming and climate change views on the greenhouse gas theory. Thomas
Allmendinger’s argument that the CO2 greenhouse gas effect is
non-existent bears serious consideration. The global warming argument
fails if CO2, a trace element in Earth’s complex atmosphere, can be
proven to have no atmosphere warming abilities not equally shared by
oxygen and nitrogen.
Read more at: ClimateDepot.com